INTERMOLECULAR FORCES
Figure 3.1 Intermolecular Forces
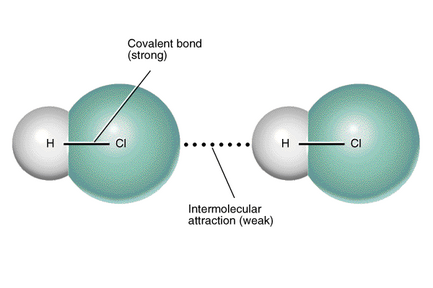
Figure 3.2 Dipole-Dipole Forces
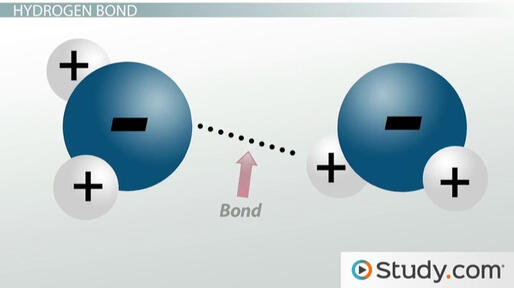
Figure 3.3 Hydrogen Bond
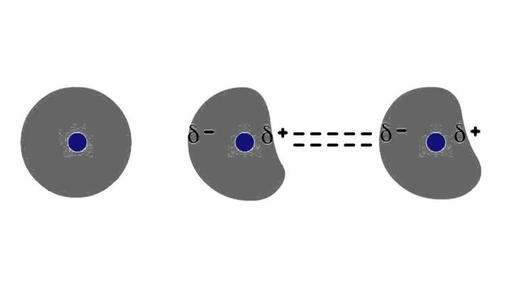
Figure 3.4 London Dispersion
What is IMFA?
The term "Intermolecular forces" is used to describe the forces of attraction between atoms, molecules, and ions when they are close to each other. When two particles interact through an intermolecular force, a positive (+) charge on one particle attracts a negative (-) charge on the other particle. This is distinct from intramolecular forces, which are another term for covalent bonds within molecules. When intermolecular forces are strong, atoms, molecules, or ions are attracted to one another and draw closer together. These are more likely to be found in condensed states like liquids or solids. When intermolecular forces are weak, atoms, molecules, or ions have little attraction for one another and move apart.
Types of Intermolecular Forces
There are three types of intermolecular forces: London dispersion forces (LDF), dipoledipole interactions, and hydrogen bonding. Molecules can have any mix of these three kinds of intermolecular forces, but all substances at least have LDF.
1. Dipole-Dipole Forces
Dipole-dipole forces exist between polar molecules—those with a permanent dipole moment due to unequal electron sharing. Because of this unequal sharing, one side of the molecule has a partial positive charge (+) and the other has a partial negative charge (-). Individual molecule polarities tend to align by opposites, drawing molecules together and favoring a condensed phase. Substances with dipole-dipole attractions have higher melting and boiling points than nonpolar molecules, which only have LDF.
2. Hydrogen Bonds
A very strong dipole is formed when a hydrogen atom is covalently bonded to nitrogen, oxygen, or fluorine. Hydrogen bonding refers to the dipole-dipole interactions that result from these dipoles. Hydrogen bonding is a particularly powerful type of dipole-dipole interaction.
3. London Dispersion Forces
LDFs exist for all substances, whether polar or nonpolar in nature. LDF are formed by the formation of temporary instantaneous polarities across a molecule caused by electron circulations. An instantaneous polarity in one molecule may induce an opposing polarity in an adjacent molecule, resulting in a series of attractive forces between neighboring molecules.
APPLICATIONS OF INTERMOLECULAR FORCES OF ATTRACTION
1. MEDICINE - IMFs are also well known for application towards prosthetic implants. Medical implants are devices that are used to replace missing body parts. It can be made from skin, body tissues, metal, plastics, or ceramics. A compound to make medical implants happen is called hydroxyapatite.

2. CONSTRUCTION MATERIALS - Construction supplies, due to the required strength from their components (sand, gravel, rocks, water, cement) have the following intermolecular forces of attraction as a consequence: ion-ion, H- bonding, dipole-dipole, ion-dipole. An example is substrates which is a material that is being glued together.

3. SPORTS EQUIPMENT - One of the most used material to build a sport equipment is Carbon Fiber. ... These apparatuses must be durable; hence the need for strong intermolecular force of attraction. › Intermolecular forces of attraction present in these materials are ion-ion, H- bonding, dipole-dipole, and ion dipole.

4. ELECTRONIC DEVISES - For electronic devices and household gadgets, dipole-induced dipole exists since components of such devices include polar substances like transient voltage suppression diodes and metalloids or semi- metals capable of having induced dipole.

https://www.britannica.com/science/gamma-decay
https://ehss.energy.gov/ohre/roadmap/achre/intro92.html
https://www.britannica.com/science/half-life-radioactivity
https://www.britannica.com/science/chemical-bonding
https://courses.lumenlearning.com/boundless-chemistry/chapter/lewis-dot-symbols-and-lewis-structures/
https://chemcenter.ucmerced.edu/sites/chemcenter.ucmerced.edu/files/page/documents/chm002workshop_imf.pdf
https://my.clevelandclinic.org/health/articles/15416-carbohydrates
https://chesquations.wordpress.com/2018/03/12/the-intermolecular-forces-of-attraction/
https://intermolecularforcessite.wordpress.com/2017/01/24/medicine/
https://chesquations.wordpress.com/
BIOMOLECULES

Figure 4.1 Carbohydrates
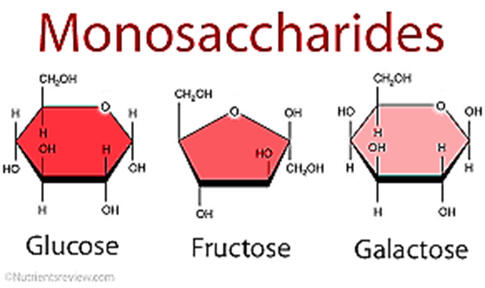
Figure 4.2 Monosaccharides
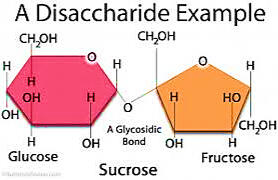
Figure 4.3 Disaccharides
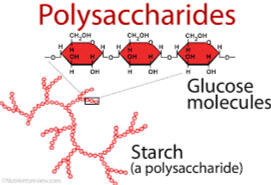
Figure 4.4 Polysaccharides

Figure 4.5 Lipids

Figure 4.6 Waxes
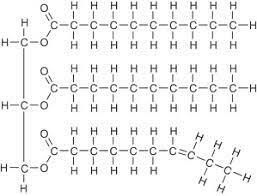
Figure 4.7 Triacylglycerol
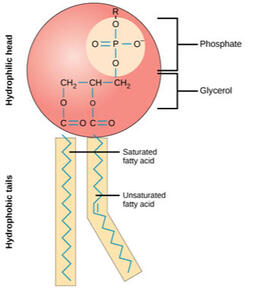
Figure 4.8 Phospolipids

Figure 4.9 Vitamins A,D,E,K

Figure 4.10 Proteins
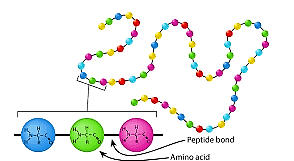
Figure 4.11 Primary
Figure 4.12 Secondary
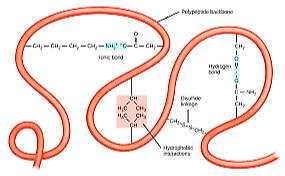
Figure 4.13 Tertiary
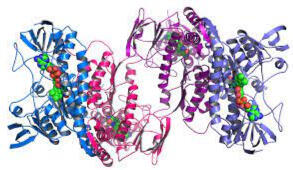
Figure 4.14
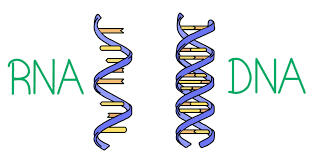
Figure 4.15 Nucleic Acid
What are Biomolecules?
These are molecules that are present in all living organism.
Biomolecules are compound of carbons. Carbohydrates (also called carbs) are a type of macronutrient found in certain foods and drinks. Sugars, starches and fiber are carbohydrates.
They are composed of subunit called monomers. Ex. Carbohydrate- glucose, Protein- amino acid.
Types of Biomolecules
1. Carbohydrates - Also known as saccharides. Its hydrate of arbon is Cn(H20)n. The primary energy source of human body with a chemical formula of CHO, 1:2:1. Refer to Figure 4.1
Functions:
Most abundant source of energy
Precursors of many organic compound (fats, amino acids)
Present as glycoprotein and glycolipids in the cell membrane
Present as structural components like cellulose in plants, exoskeleton in some insects, and cell wall of microorganisms
Storage form of energy (glycogen) to meet the energy demands of the body.
Types of Carbohydrates
A. MONO SACCHARIDE - The simplest sugar. The basic sub unit of .carbohydrate. C6H12O6. Refer to the Figure 4.2
• Glucose – used in dextrose, blood sugar
• Fructose – found in fruit and honey
• Galactose – found in milk and milk product
B. DI SACCHARIDE - Consist of 2 monosaccharide. Bonded by glycosidic bond. They are crystalline, water soluble and sweet to taste. Refer to the Figure 4.3
• Maltose- malt sugar, made up of 2 glucose
• Lactose- milk sugar, made up of glucose and galactose
• Sucrose- cane sugar, made up of glucose and fructose
C. POLY SACCHARIDE
Complex sugar. Used as source of energy and structure. Allow the organism to allow the use of energy gradually since it is stored in long structure. Refer to the Figure 4.4
• Starch – energy storage in plants
• Glycogen – energy storage in animals
• Cellulose – part of the cell wall of plants
2. Lipids - It is any of a class of organic compounds that are fatty acids or their derivatives and are insoluble in water but soluble in organic solvents. They include many natural oils, waxes, and steroids. Refer to the Figure 4.5
• Contain large number of non polar carbon-carbon and carbon-hydrogen bonds.
• Insoluble with water
• Soluble in ether, chloroform, acetone and benzene
• Made of fatty acids
Classifications of Lipids
A. Hydrolyzable - It can be converted into smaller molecules by hydrolysis with water.
• WAXES - They are formed from fatty acids and high molecular weight alcohol. Their long non polar carbon chain makes them very hydrophobic. (Di sila nagbeblend w/ water). Figure 4.6
• TRIACYLGLYCEROL - Triacylglycerol or triglyceride are trimesters formed from glycerol and three molecules of fatty acids. Fats and oil are triacylglycerols with different physical properties. Figure 4.7
• PHOSPHOLIPIDS - Lipids that contain phosphorus atom. Makes the cell membrane. Figure 4.8
B. Nonhydrolyzable - They cannot be broken down into smaller unit by aqueous hydrolysis. They tend to be varied in structure.
• STEROID - Biologically active steroids are hormones secreted by the endocrine glands. There are two types of steroid hormone the sex hormones and adrenal cortical steroid.
SEX HORMONES FEMALE
Estrogen
Progesterone
SEX HORMONES MALE
Androgen
Adrenal Cortical Steroid - Synthesized in the outer layer of the adrenal gland. It includes aldosterone, cortisone and cortisol.
• FAT SOLUBLE VITAMINS - Organic compounds requires in small quantities for normal metabolism. They cannot be synthesized by our cells and fat soluble vitamins are vitamins dissolve in lipids which are the vitamins ADEK. Refer to the Figure 4.9
Vitamin A - Responsible for healthy eye sight and mucus membrane.
Vitamin D - Helps in regulation in the metabolism of both phosphorus and calcium in the bone.
Vitamin E - Anti oxidant and it protect unsaturated side chains in fatty acids from unwanted oxidation.
Vitamin K - Regulates the synthesis of prothrombin and other proteins needed for blood to clot.
• EICOSANOID - Potent compound present in low concentration in cells. They are synthesize from arachidonic acid in response to external stimulus. They are called local mediator meaning they work in the environment which they are synthesized.
Prostaglandins - They are responsible for inflammation. Decrease gastric secretions. Inhibit blood platelet aggregation
Stimulate uterine contraction.
Leukotrienes
Molecules that contribute to the asthmatic response by constricting smooth muscles especially in lungs.
3. Proteins - It contains amide bonds, formed by joining amino acid together. Amino acid is the monomer of protein and it is composed of CHON.
FUNCTIONS:
Provides support and structure of the body
Regulates body’s metabolism
Helps in muscle movement
Fights infections
AMINO ACIDS
All amino acids include five basic parts:
a central carbon atom.a hydrogen atom.an amino groupconsisting of a nitrogen atom and two hydrogen atoms.a carboxyl group - consisting of a carbon atom, two oxygen atoms, and one hydrogen atom.an R-group or side chain - consisting of varying atoms.
Protein Four Levels of Structure
PRIMARY - Figure 4.11
• The simplest structure of amino acids
• Sequence of amino acid in polypeptide chain
• The linear sequence of amino acids
SECONDARY - Figure 4.12
• the localized organization of parts of a polypeptide chain
•
TERTIARY - Figure 4.13
• Three dimensional arrangement of the polypeptide chain
QUARTERNARY - Figure 4.14
• the association of two or more polypeptides into a multi subunit complex
4. Nucleic Acid - Act as storage of genetic information. It has nucleotides as monomer. It also has two classifications namely: Deoxyribonucleic Acid (DNA) and Ribonucleic Acid (RNA).
Nucleotides are composed of...
1. Sugar
2. Phosphate
3. Nitrogenous bases
FUNCTIONS:
DNA directs protein synthesis
RNA helps in synthesis of protein
2 types mRNA and tRNA


